BEVEL
BEVEL is a compact, portable autotransfusion device originally designed to recover and reinfuse a patient's own blood during postpartum hemorrhage; one of the leading causes of maternal death worldwide.
We partnered with BAKS Medical to help them answer a critical question: who needs this device, and how do you get it to them?
What started as a maternal health challenge quickly evolved into something much larger, as our research uncovered an opportunity to expand BEVEL's reach far beyond the delivery room.
October 2024
[Research heavy · 10 min read]
CLIENT
BAKS Medical
ROLE
User Researcher
Product Designer
Strategist
TOOLS
Mural
Figma
Slack
Google Workspace
_____ Background
What we did
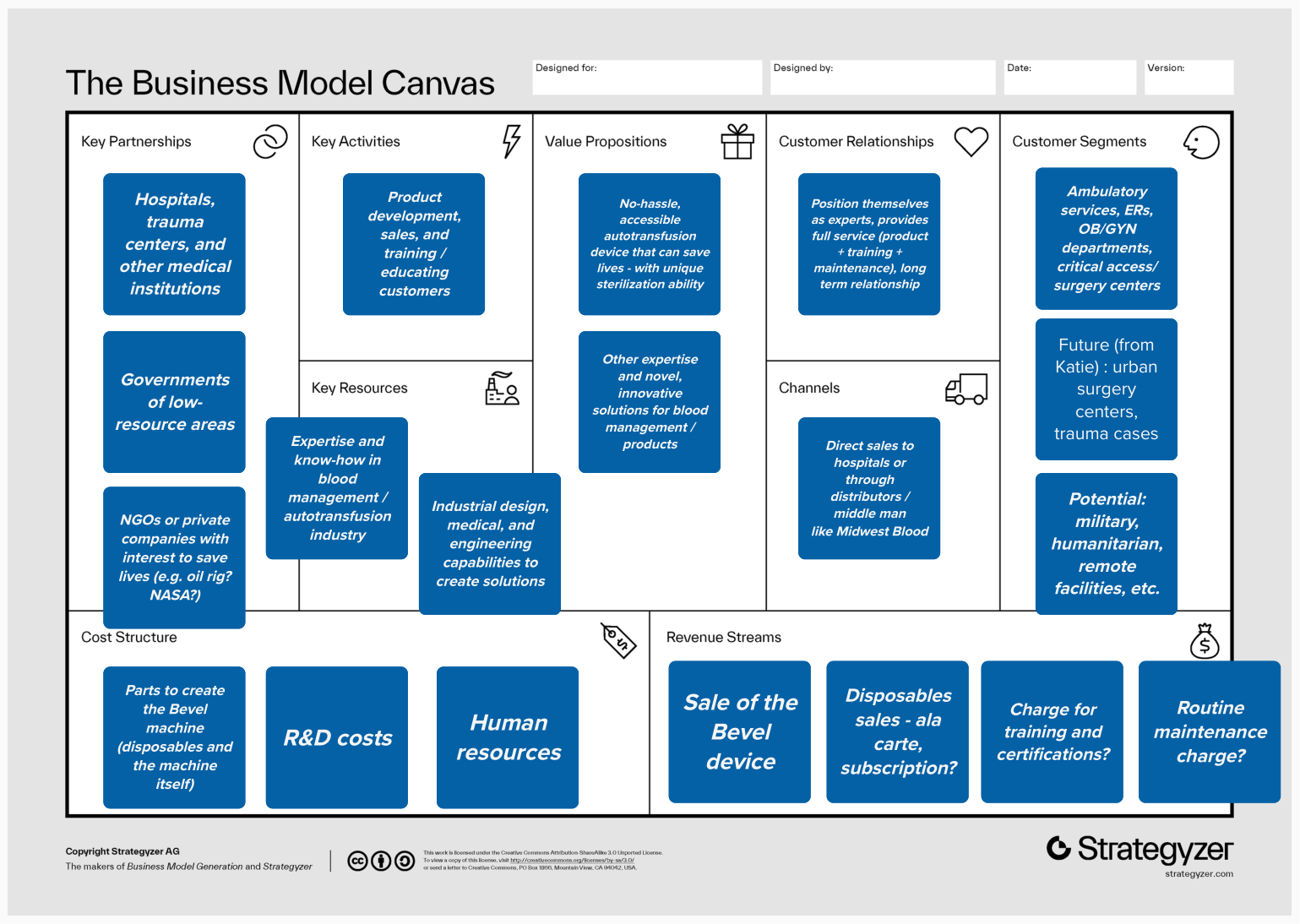
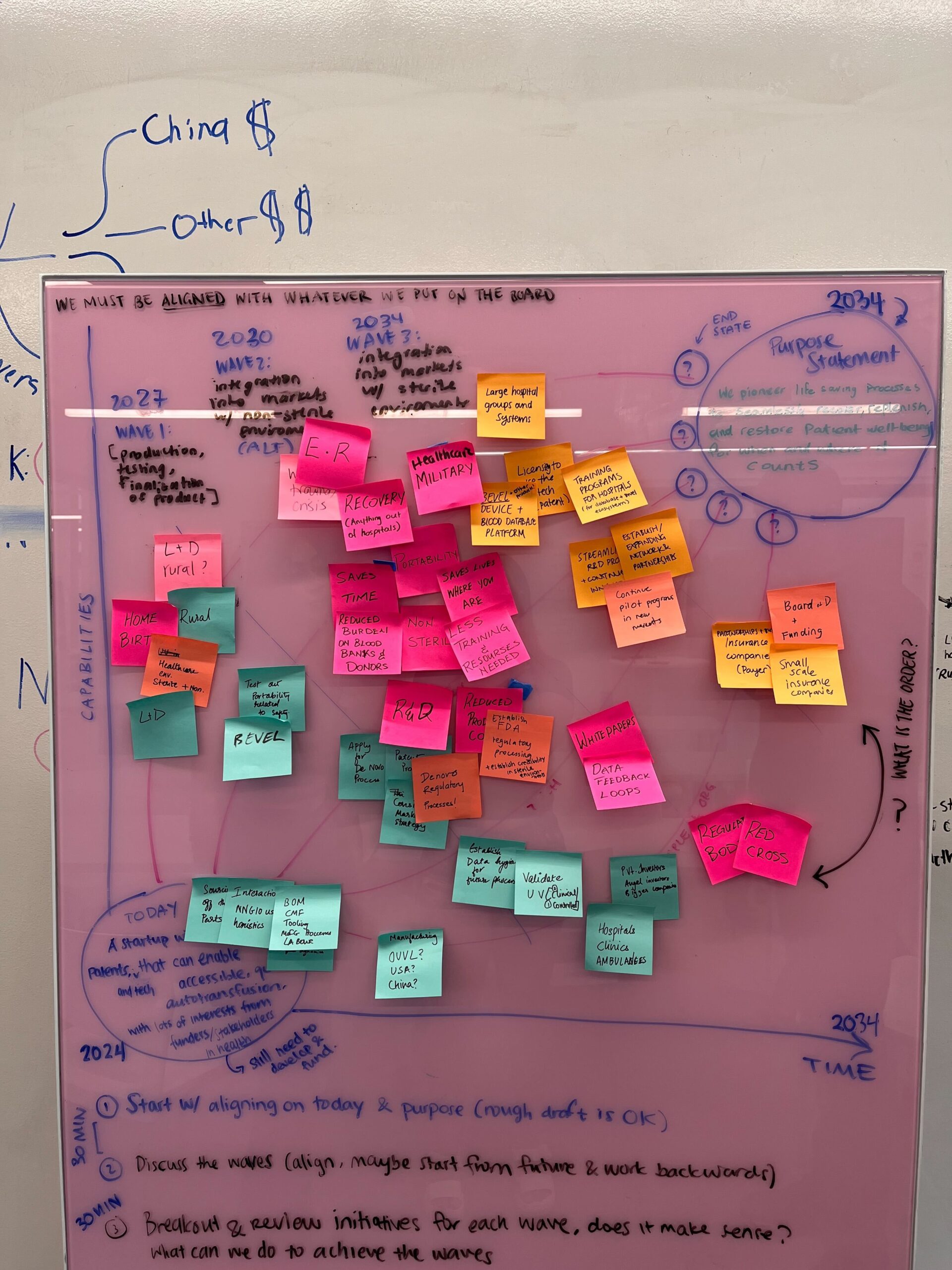
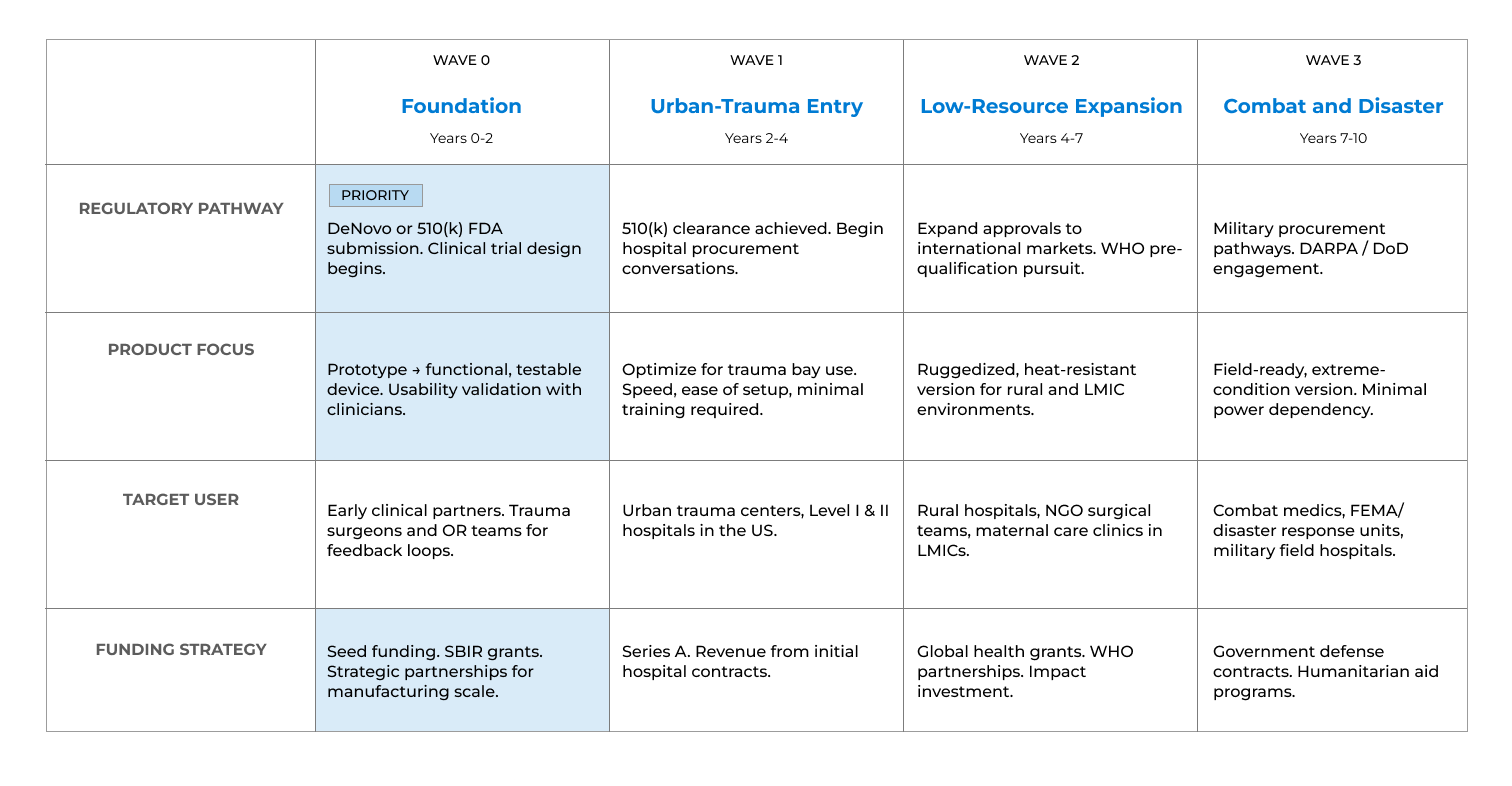
We were brought in expecting to focus exclusively on the maternal health market, the problem BEVEL was originally designed to solve. But through 6+ in-depth interviews with clinicians, surgeons, and subject matter experts, our research pointed us somewhere bigger. We identified not one but three distinct markets for BEVEL and built a four-wave, 10-year strategic roadmap to get there. We developed user personas, mapped clinical workflows, and translated field insights into a go-to-market strategy that repositioned BEVEL from a maternal device into a life-saving platform with far broader reach.
How we did it
Using the Resonance Framework in week one, we aligned with BAKS Medical on their mission and constraints. We then conducted secondary research into the autotransfusion landscape, competitor devices, and FDA regulatory pathways. In weeks four and five, we conducted qualitative interviews with an industrial designer involved in BEVEL's early development, an emergency care physician with direct trauma experience, and three SMEs across medical devices, regulation, and surgical environments.
The patterns that emerged challenged our original brief
and redirected the entire strategy.
14 mil
women affected by
postpartum hemorrhage annually
50 %
of surgeries in low-income countries lack adequate blood access
90 %
of combat deaths happen before reaching care
BEVEL was designed for one room.
Our research found it was needed everywhere.
_____ Work Highlight
Challenging the brief
The original scope pointed us toward one market. We let the research speak louder.
What we found completely reframed how BEVEL could be positioned, who it could serve, and where it could go next.
Bridging Research and Reality
By speaking directly with people on the frontlines; from emergency physicians to critical care workers, we translated lived clinical experience into a strategy grounded in actual need, and not just and ideal path.
_____ Conclusion
Future of Bevel
What started as a maternal health project became a $390M market opportunity.
Our research didn't just validate BEVEL; it expanded its entire vision. By listening to the people on the frontlines of blood loss emergencies, we gave BAKS Medical a clear, phased roadmap: enter urban trauma first, expand to rural settings, and ultimately serve combat medics and disaster relief teams around the world.
_____ Research
Week 1
We began by understanding BAKS Medical’s mission, vision, and expectations. Through team workshops and discussions with BAK's medical team, we aligned on a shared purpose and laid the foundation for meaningful collaboration.
Used Resonance Framework
Week 2-3
We conducted secondary research to explore the autotransfusion space, competitors, and regulatory pathways.

How is BEVEL different than other blood autotransfusion devices?
Portable
Compact
UV Sterile
Week 4-5
Through interviews gained deep insights into user needs, product fit, and potential market positioning.
This helped us validate assumptions and identify gaps in the current market.
We Conducted Interviews With
1. An industrial designer involved in BEVEL’s early development.
2. An emergency care physician with direct experience in trauma scenarios.
3. Three subject matter experts with backgrounds in medical devices, regulation, and surgical environments.
Who is your target customer?
What is your value proposition?
How will you deliver?
How will you make money?
Week 6-7
Based on our research, we developed a four-phase strategic roadmap and launch strategy. We concluded with a final presentation to stakeholders, outlining key recommendations for product development, funding, and go-to-market planning.

United States
Email: sampadapote1@gmail.com
© Sampada Pote 2025



